🔐 Secure Payment Guaranteed
Safe checkout with trusted global payment methods.
🌟 Why Choose Infinity Market Research?
At Infinity Market Research, we dont just deliver data — we deliver clarity, confidence, and competitive edge.
In a world driven by insights, we help businesses unlock the infinite potential of informed decisions.
Here why global brands, startups, and decision-makers choose us:
Industry-Centric Expertise
With deep domain knowledge across sectors — from healthcare and technology to manufacturing and consumer goods — our team delivers insights that matter.
Custom Research, Not Cookie-Cutter Reports
Every business is unique, and so are its challenges. Thats why we tailor our research to your specific goals, offering solutions that are actionable, relevant, and reliable.
Data You Can Trust
Our research methodology is rigorous, transparent, and validated at every step. We believe in delivering not just numbers, but numbers that drive real impact.
Client-Centric Approach
Your success is our priority. From first contact to final delivery, our team is responsive, collaborative, and committed to your goals — because you re more than a client; you re a partner.
Recent Reports
AI Motion Capture Software Market
Chlorinated Methoxy Fatty Acid Methyl Ester Market
Global Touch Activated Phlebotomy Device Market Size, Share and Analysis Report 2026-2032
Mar 2026
Healthcare
Pages: 105
ILR4738
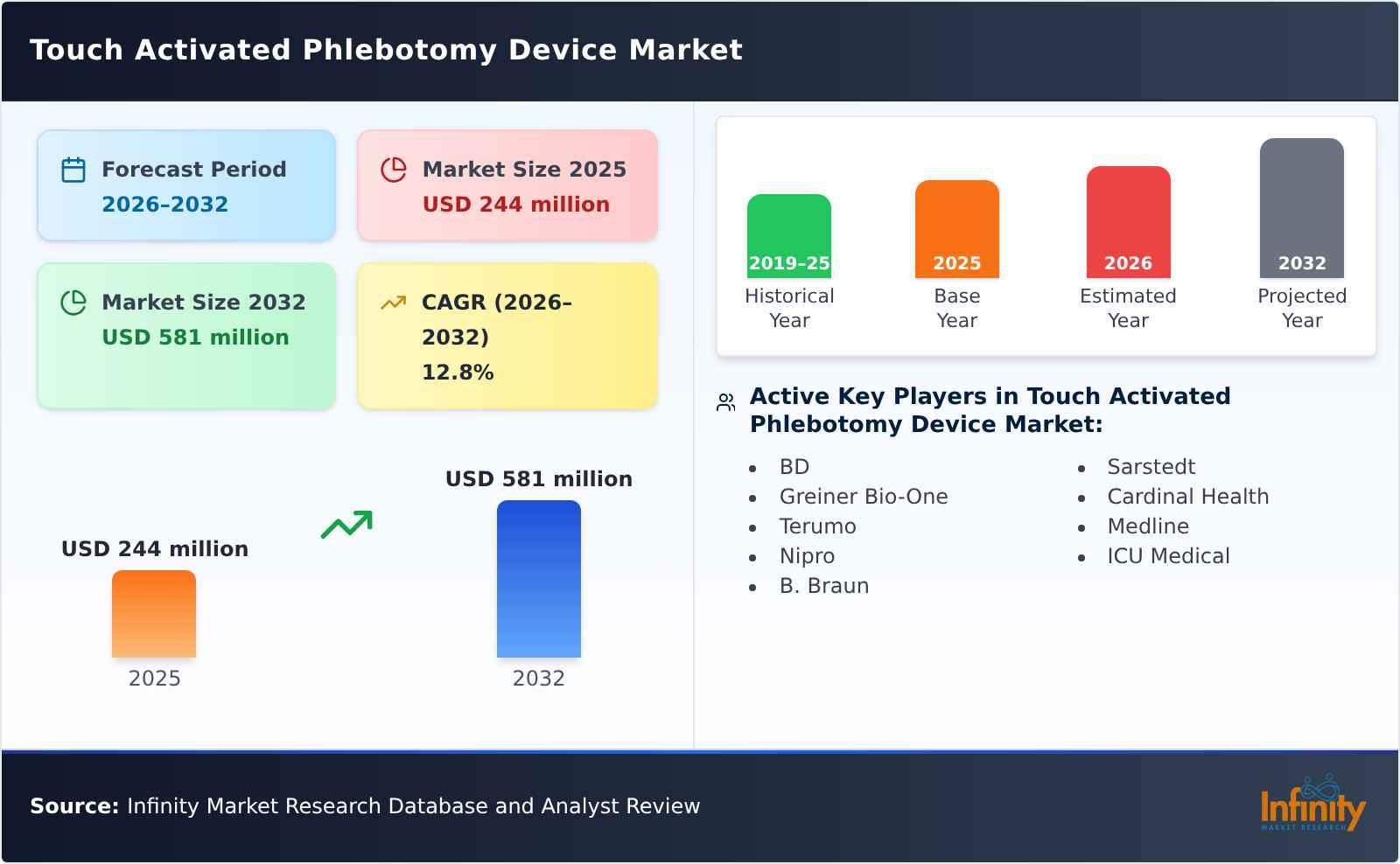
The global Touch Activated Phlebotomy Device market size is predicted to grow from US$ 244 million in 2025 to US$ 581 million in 2032; it is expected to grow at a CAGR of 12.8% from 2026 to 2032.
Explore this report in detail? Download a free sample copy
Download Free Sample Report
Touch Activated Phlebotomy Device refers to a venous blood collection device engineered around touch-triggered, often one-hand activation. It typically integrates needle protection, puncture activation, blood return indication/feedback, hemostasis support, and safety lockout in a single system to reduce procedural steps while improving consistency and safety. The device can be configured for vacuum tube collection, butterfly sets/extension tubing, or dedicated connector interfaces, serving routine laboratory phlebotomy and selected bedside sampling workflows. Its core value lies in touch-activated mechanics and closed or semi-closed fluid pathways that minimize exposed needle time and blood splashes, lowering needlestick risk, improving patient comfort, and enabling more standardized, efficient, and compliant phlebotomy operations.In 2025, global Touch Activated Phlebotomy Deviceproduction reached approximately 24.9 million units and price is about 10 usd/unit.The average gross profit margin of this product is 55%.
As healthcare providers strengthen occupational exposure prevention, infection control, and procedural standardization, safety upgrades in phlebotomy have become a firm requirement. Touch-activated phlebotomy devices align with the need to reduce steps, minimize exposure, and improve consistency?especially in high-throughput settings such as emergency departments, outpatient draw stations, and centralized pre-analytical workflows. Workforce shifts and rising training costs further favor intuitive, repeatable devices that reduce dependence on individual technique. In many institutions, ?safe phlebotomy? is evolving into a system-level procurement and governance theme, driving upgrades from single consumables toward integrated offerings that combine devices, compatible disposables, and service?expanding the commercial runway for suppliers with interface ecosystems and scalable delivery capabilities.
Challenges center on clinical adaptability, cost sensitivity, and compliance validation. Venous variability, insertion angle, and activation force can influence first-stick success and patient experience; the device must deliver stable performance across patient populations and departments. Any issues such as insensitive triggering, unclear blood return feedback, or connector incompatibility can increase procedure time and complaints. Phlebotomy supplies are also high-frequency and cost-sensitive, so suppliers must balance safety upgrades with tight cost control and reliable supply. In addition, products incorporating needle safety mechanisms require robust quality systems, regulatory compliance, and real-world usability validation; inadequate training or inconsistent use can blunt the intended safety benefits and undermine repurchase and adoption within the hospital.
Demand is shifting from ?usable? to ?controllable, traceable, and standardized.? Providers increasingly focus on the entire pre-analytical quality chain?patient identification, collection consistency, hemolysis/clotting control, sample logistics efficiency, and documentation of occupational exposure events. Therefore, touch-activated devices must not only be safe and easy to use, but also fit seamlessly into vacuum tube systems, phlebotomy carts, and specimen management workflows. Health-check centers and IVD-related service settings emphasize throughput and low error rates, favoring solutions that shorten cycle time and reduce rework. For populations with difficult venous access (pediatrics, geriatrics, oncology), the market leans toward variants with enhanced control and clear visual/functional feedback. Overall, product portfolios are evolving toward scenario-specific series and a complementary consumable ecosystem.
Key upstream inputs include medical-grade stainless-steel cannulae and bevel grinding, medical polymers for hubs/housings and activation structures, elastomers and seals for valves and leak prevention, transparent materials for observation windows, and connector components compatible with vacuum tubes or extension sets. For safety lockout and anti-rebound mechanisms, consistency of micro-springs, latches, and precision assembly determines activation feel and reliability. Because the device contacts blood directly or operates in near-blood environments, biocompatibility, sterilization compatibility, extractables/leachables control, and batch stability are critical. At scale, injection mold accuracy, automated assembly yield, and in-line inspection capabilities become decisive. Ultimately, upstream stability and quality management translate into first-stick performance, leakage risk, and safety-mechanism reliability?forming the foundation for brand trust and long-term institutional partnerships.
Infinity Market Research newest research report, the ?Touch Activated Phlebotomy Device Industry Forecast? looks at past sales and reviews total world Touch Activated Phlebotomy Device sales in 2025, providing a comprehensive analysis by region and market sector of projected Touch Activated Phlebotomy Device sales for 2026 through 2032. With Touch Activated Phlebotomy Device sales broken down by region, market sector and sub-sector, this report provides a detailed analysis in US$ millions of the world Touch Activated Phlebotomy Device industry.
This Insight Report provides a comprehensive analysis of the global Touch Activated Phlebotomy Device landscape and highlights key trends related to product segmentation, company formation, revenue, and market share, latest development, and M&A activity. This report also analyzes the strategies of leading global companies with a focus on Touch Activated Phlebotomy Device portfolios and capabilities, market entry strategies, market positions, and geographic footprints, to better understand these firms? unique position in an accelerating global Touch Activated Phlebotomy Device market.
This Insight Report evaluates the key market trends, drivers, and affecting factors shaping the global outlook for Touch Activated Phlebotomy Device and breaks down the forecast by Type, by Application, geography, and market size to highlight emerging pockets of opportunity. With a transparent methodology based on hundreds of bottom-up qualitative and quantitative market inputs, this study forecast offers a highly nuanced view of the current state and future trajectory in the global Touch Activated Phlebotomy Device.
This report presents a comprehensive overview, market shares, and growth opportunities of Touch Activated Phlebotomy Device market by product type, application, key manufacturers and key regions and countries.
Segmentation by Type:
Peripheral Venous Phlebotomy
Central Line Sampling Support
Other
Segmentation by Needlestick:
Passive Safety
Active Safet
Segmentation by Safety Mechanism:
Passive/Auto-Engage
User-Activated Mechanisms
Other
Segmentation by Application:
Hospitals
Diagnostics/Reference Labs
Other
This report also splits the market by region:
Americas
United States
Canada
Mexico
Brazil
APAC
China
Japan
Korea
Southeast Asia
India
Australia
Europe
Germany
France
UK
Italy
Russia
Middle East & Africa
Egypt
South Africa
Israel
Turkey
GCC Countries
The below companies that are profiled have been selected based on inputs gathered from primary experts and analysing the companys coverage, product portfolio, its market penetration.
BD
Greiner Bio-One
Terumo
Nipro
B. Braun
Sarstedt
Cardinal Health
Medline
ICU Medical
Key Questions Addressed in this Report
What is the 10-year outlook for the global Touch Activated Phlebotomy Device market?
What factors are driving Touch Activated Phlebotomy Device market growth, globally and by region?
Which technologies are poised for the fastest growth by market and region?
How do Touch Activated Phlebotomy Device market opportunities vary by end market size?
How does Touch Activated Phlebotomy Device break out by Type, by Application?
Touch Activated Phlebotomy Device Market Scope
| Report Attribute | Details |
|---|---|
| Market Size (Start Year) | USD XX Million |
| Market Size (End Year) | USD XX Million |
| Compound Annual Growth Rate (CAGR) | USD XX Million |
| Forecast Period | USD XX Million |
| Base Year | USD XX Million |
| Historical Data | USD XX Million |
| Key Players | USD XX Million |
REPORT COVERAGE
Revenue forecast, Company Analysis, Industry landscape, Growth factors, and Trends
SEGMENT COVERED
By component, deployment, organization size, application, and industry.
REGIONAL SCOPE
North America, Europe, Asia Pacific, Middle East & Africa, South & Central America
COUNTRY SCOPE
Includes key countries across all major regions.
📘 Frequently Asked Questions
1. What is the market size of Global Touch Activated Phlebotomy Device Market?
Answer: The global Touch Activated Phlebotomy Device market size is predicted to grow from US$ 244 million in 2025 to US$ 581 million in 2032; it is expected to grow at a CAGR of 12.8% from 2026 to 2032.
2. Which regions are analyzed in the Global Touch Activated Phlebotomy Device Market report?
Answer: The Global Touch Activated Phlebotomy Device Market report covers major regions such as Europe, Middle East & Africa. Each region is analyzed for trends, opportunities, and market dynamics.
3. What methodology is used for forecasting of Global Touch Activated Phlebotomy Device Market?
Answer: The Global Touch Activated Phlebotomy Device Market report uses a mix of primary research, secondary data, and expert analysis to build its forecasts. Models include both qualitative and quantitative approaches.
4. Are emerging markets analyzed separately in the Global Touch Activated Phlebotomy Device Market?
Answer: Yes, the Global Touch Activated Phlebotomy Device Market report highlights high-growth emerging regions with dedicated insights. These include untapped opportunities, risks, and potential for expansion.
5. Does the report include competitive benchmarking of Global Touch Activated Phlebotomy Device Market?
Answer: Yes, Global Touch Activated Phlebotomy Device Market report compares major players based on revenue, product portfolio, innovation, and regional presence. This helps assess competitive positioning.
6. Can I access country-level data within the Global Touch Activated Phlebotomy Device Market report?
Answer: Yes, Global Touch Activated Phlebotomy Device Market report includes detailed data by country, especially for key markets. This allows for localized insights and decision-making.
7. Can I get customized insights or data from the Global Touch Activated Phlebotomy Device Market report?
Answer: Yes, we offer customization options to align with your specific business needs. You can request tailored sections or regional breakdowns.
🔐 Secure Payment Guaranteed
Safe checkout with trusted global payment methods.
🌟 Why Choose Infinity Market Research?
At Infinity Market Research, we dont just deliver data — we deliver clarity, confidence, and competitive edge.
In a world driven by insights, we help businesses unlock the infinite potential of informed decisions.
Here why global brands, startups, and decision-makers choose us:
Industry-Centric Expertise
With deep domain knowledge across sectors — from healthcare and technology to manufacturing and consumer goods — our team delivers insights that matter.
Custom Research, Not Cookie-Cutter Reports
Every business is unique, and so are its challenges. Thats why we tailor our research to your specific goals, offering solutions that are actionable, relevant, and reliable.
Data You Can Trust
Our research methodology is rigorous, transparent, and validated at every step. We believe in delivering not just numbers, but numbers that drive real impact.
Client-Centric Approach
Your success is our priority. From first contact to final delivery, our team is responsive, collaborative, and committed to your goals — because you re more than a client; you re a partner.


